Feb 07, 2017
Philips and Diagnostics Development win European Union ‘Horizon Prize – Better Use of Antibiotics’ for the rapid detection of bacterial infection on Philips’ Minicare I-20 platform
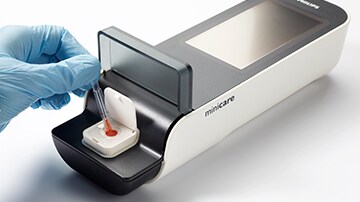
Antimicrobial agents – such as antibiotics – have dramatically reduced the number of deaths from infectious diseases since their introduction 70 years ago. However, through their overuse and misuse, many micro-organisms have become resistant to them. It is estimated that each year this growing antimicrobial resistance (AMR) causes around 25,000 deaths and accounts for over EUR 1.5 billion in healthcare costs and productivity losses in Europe alone, making it one of the greatest challenges facing society today. To boost development and bring new and innovative players to AMR research, the EU issued a prize for the individual or team that most effectively meets the following challenge: ”to develop a rapid test that will allow healthcare providers to distinguish at the point of care between patients with upper respiratory tract infections that require antibiotics and those that can be treated safely without antibiotics”. “This prize is a great recognition of how collaboration can lead to innovative diagnostic solutions that improve patient care,” said Marcel van Kasteel, CEO of Handheld Diagnostics at Philips. “The HNL program for detection of bacterial infections perfectly fits with our ‘open platform’ strategy of bringing innovative biomarker content onto the Minicare I-20 platform in order to offer healthcare professionals a panel of relevant tests for acute settings. Activities are ongoing to further develop the HNL test for eventual implementation in routine clinical practice. “In various studies, HNL has demonstrated superior performance compared to biomarkers currently used to diagnose bacterial infection,” said Per Venge, CEO of P&M Venge AB. “I am therefore very excited that we have been able to successfully demonstrate the detection of HNL on the Minicare I-20 using blood samples from patients with upper respiratory tract infections in the Uppsala University hospital, where within a few minutes patients with a bacterial infection could clearly be distinguished from those with viral infections or other conditions.” Based on Philips’ proprietary biosensor technology, the Minicare I-20 handheld diagnostics platform is designed to detect multiple target molecules at very low concentrations in a single ‘finger-prick’ blood sample, and display the results on a handheld analyzer within minutes. Philips already has CE marking for its Minicare I-20 based cardiac troponin I (cTnI) blood test for the rapid point-of-care diagnosis of heart attack, and it is currently developing further tests for acute care settings. Minicare I-20 is simple and easy to use by non-laboratory staff. The analyzer’s built-in connectivity allows direct transfer of the data to laboratory and hospital information systems to update patient files, while integrated calibration and fail-safe functionalities ensure the robustness and accuracy needed for confident on-the-spot decision making.
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people's health and enabling better outcomes across the health continuum from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. The company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Headquartered in the Netherlands, Philips' health technology portfolio generated 2016 sales of EUR 17.4 billion and employs approximately 71,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.